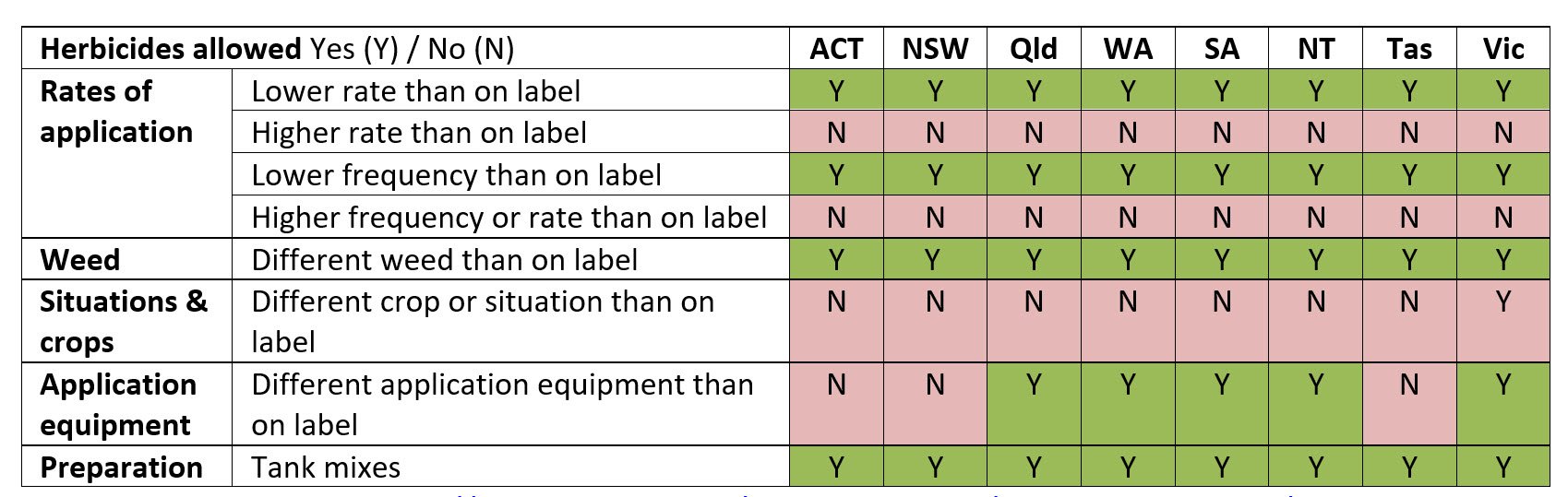
Pesticide use in Australia varies between the states as each state has responsibility and develops the regulations for how they are used. Some commonalities of use evolved during the early 2000s, but it is only recently that all states and territories have mostly brought off-label uses in line with each other as seen in Table 1.
What is off-label use?
Off-label use is the use of an agricultural chemical to address a specific issue that is not covered by the APVMA approved label, or permit issued to ‘persons generally’, excluding vertebrate poisons e.g. 1080.
Note: Victoria alone also has a state-based permit system as well as the APVMA permit system.
Wilful misuse on the other hand represents an active or negligent disregard for all instructions and legal requirements with no consideration of the risks.
Table 1. State Control of use legislation (off-label) in agricultural chemicals (as of May 2022) For more information: https://www.awe.gov.au/agriculture-land/farm-food-drought/ag-vet-chemicals/domestic-policy/haccut
Warning!
Off-label practices do not exclude or override product maximum residue limits, work health and safety or environmental safety. If going off-label the decisions the user must consider include the rate of pesticide; time and frequency of application; the likelihood of residues; and the potential for worker exposure.
‘Off-label use’ does not override Directions for Use ‘DO NOT’ statements on labels and permits such as “DO NOT apply to crops or pastures with clover, lucerne or medics”.
When would I go “off-label”?
Different weed to what is on the label
It is likely the most common “off-label” use would be to treat a weed species that is not on a current herbicide label or permit.
Firstly check whether there is a registration or permit for that weed by going to the APVMA web site and check ‘PubCRIS’ database for registrations and the Permit database for labels. https://apvma.gov.au/node/10831
Sea spurge (Cakile maritima) Port Fairy, Victoria
Only go off-label if there are closely related species on the herbicide label or there is trial data proving how effective the herbicide and rate is on the target species. For example sea rocket (Cakile maritima) is a widespread weed of the southern Australian coast, however there are no herbicides registered for its control and no APVMA permits exist. Being a brassica, it is likely herbicides registered for the control of brassicas weeds such as wild radish (Raphanus raphanistrum) would also be effective.
If we look at 2,4-D amine (625 g/L) there are a wide range of brassica species already on the label. However label restrictions might limit off-label use. For example:
· DO NOT apply by spray equipment carried on the back of the user i.e. a pressurised backpack sprayer
· Downwind buffer zones for natural aquatic areas
· Must be applied as spray droplets no smaller than a VERY COARSE spray quality.
· Some individual state restrictions might also apply.
Different crop or situation on the label
Only Victoria allows this. The principal reason is that there might be issues with residues in the end product if there are no maximum residue limits set for that active ingredient in that crop/product.
Different application equipment than on the label
This is not allowed in the ACT, NSW and Tasmania.
If you wanted to use different application equipment in those places you would need to apply for an APVMA Permit. However this does not override statements on the label. Using the 2,4-D amine (625 g/L) label as an example you still cannot:
use a backpack sprayer
use a vertical sprayer
use equipment that produces a spray quality finer than VERY COARSE
Tank Mixes
Off-label tank mixes are allowed in all states and territories. However tank mix components used in a situation must:
be registered/permitted for that situation
they must be compatible in the tank - for example mixing a grass selective herbicide such as Fusilade® Forte with 2,4-D will reduce the level of grass control. In other situations mixing different formulations causes a precipitation of the herbicide to the bottom of the tank.
biologically compatible in the weed – when targeting sowthistle (Sonchus spp.) mixing glyphosate with 2,4-D reduces the level of control due to the effect the herbicides have on the sowthistle’s biochemical processes